The Cost of Overactive Immunity
Written by Andrea Mendez Aguirre
Edited by Raghav Tewari
Why was Covid 19 so deadly? Why was it that people often passed away even weeks after the infection had cleared? To answer this, we must analyze one of the body´s most important protective mechanisms: immunity.
In broad terms, the immune system is activated when immune cells encounter an element recognized as ´not self´. This event then triggers two highly intertwined mechanisms. The first to act is the innate, or non-specific, immune response, which acts quickly to fight off the germ through a variety of strategies, including mucous membranes, proteins, and specialized cells such as phagocytes. However, when this response fails to eliminate the invader, the body switches to the adaptive, or specialized, immune response, which specifically targets the germ causing the infection. Although slower, it is far more effective, and it is also why we develop immunity to certain germs after being infected [1].
Photo from Getty Images. Curated by Andrea Mendez Aguirre (am3362@cornell.edu).
The action of the immune system, however, is subject to a careful balance that, if disturbed, can lead to devastating consequences. One element that must remain balanced is the ratio of pro- and anti-inflammatory cytokines, which are important regulatory signaling proteins produced by both the innate and adaptive immune response [2]. The COVID-19 example is of great interest here, as affected patients have been reported to show elevated levels of pro-inflammatory cytokines. This observation also seems to correlate with disease severity [3]. Such a phenomenon is often referred to as a cytokine storm, and it explains the disproportionate inflammatory response seen in patients manifested as respiratory distress syndrome and organ failure - the actual main causes of death during the pandemic [4]. This is notably different from other infectious diseases, in which the virus itself destroys cells, causing primary damage.
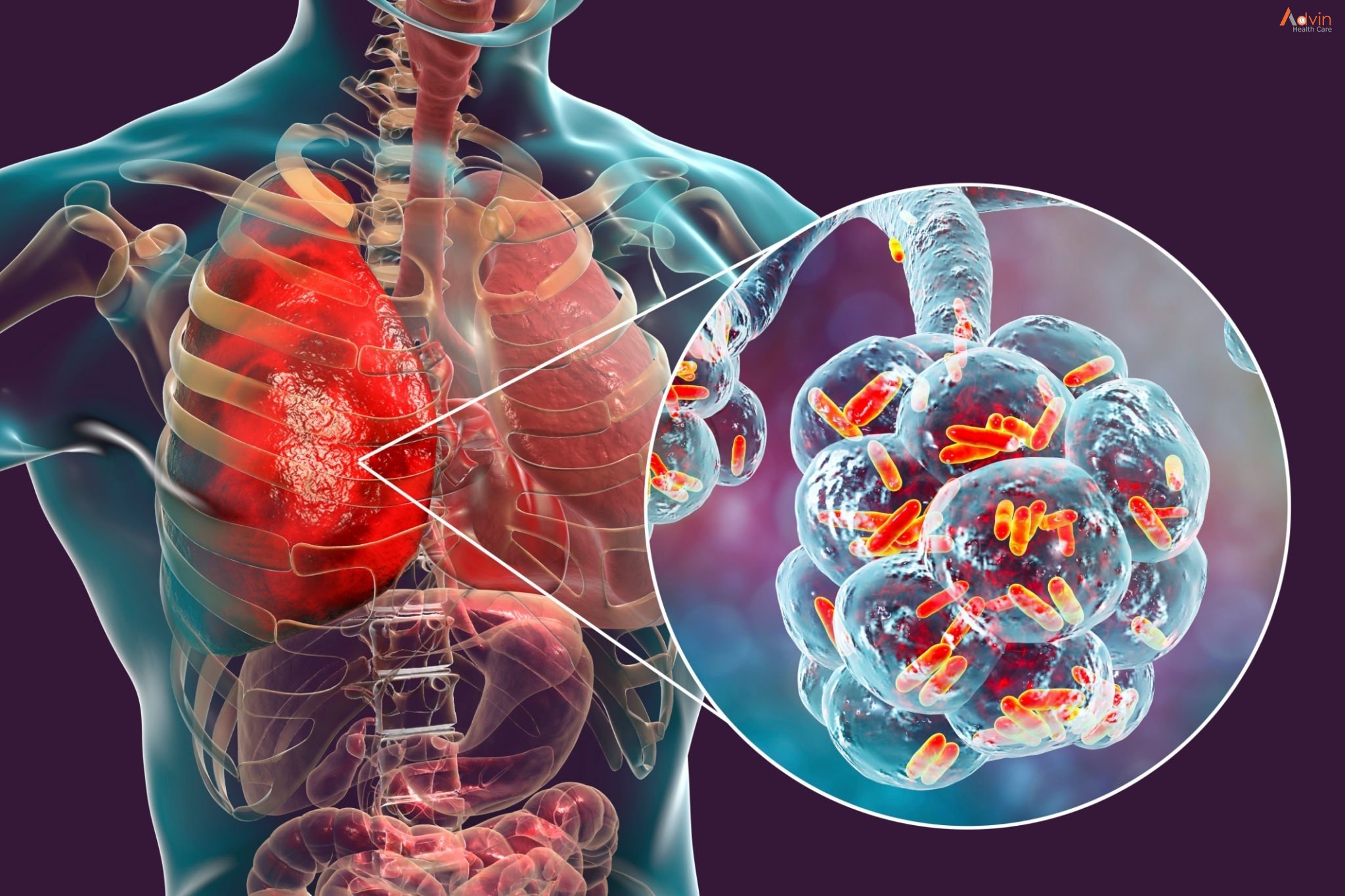
Another great example of exacerbated immune response is Tuberculosis. Unlike SARS-CoV-2 (a virus), the causative agent of tuberculosis is the bacterium M. tuberculosis. When M. tuberculosis infects a host, it is ingested by macrophages and sequestered by other immune cells to fight off the pathogen, forming a structure called a granuloma. Although the granuloma contains the infection and prevents the host from showing symptoms, it is also, unfortunately, an ideal environment for the bacteria to proliferate and survive for decades. The immune cells in the granuloma will eventually die, a process called necrosis. This leads to the development of the granuloma into its caseous, or disintegrating, stage, which propels the disease into its lethally symptomatic, active, and lung-destroying form [5]. It is almost ironic that in an attempt to resist infection, the immune system inadvertently creates the optimal conditions necessary for disease progression.
Photo from Advin Healthcare. Curated by Andrea Mendez Aguirre (am3362@cornell.edu).
The immune system is far more complex than simply killing germs and pathogens that infect us. It is neither a hero nor a villain. Just as it is capable of healing, it is also capable of harming. Understanding when protection becomes pathology is a central question and one of the most important challenges in modern medicine. It is no longer just about stimulating immunity, but about tuning it to optimum conditions. For example, corticosteroids [6] and other immune-regulator drugs, such as TNF and JAK inhibitors, represent an advancement into a new era of medicine that seeks to recalibrate the immune system. Instead of fighting the infectious agent directly, as more common drugs do, they focus on regulating the immune response. In doing so, they confront one of biology´s central paradoxes: that the very force designed to protect us can, when unchecked, become the source of its greatest harm.
Andrea Mendez Aguirre ‘28 is in the College of Agriculture and Life Sciences, studying Biological Sciences. She can be reached at am3362@cornell.edu.