Why Does Biological Resilience Decline After Childhood?
Written by Rathusha Nimalan
Edited by Gesi Huang
The human body is remarkably adaptable for most of one’s early life. Children generally recover quicker from infections and injuries than adults and adjust to new environments with greater flexibility. The ability of kids to “bounce back” is a direct result of the ability of their physiological systems to restore stability after stress. However, as humans age beyond childhood, this resilience markedly declines. By mid-life, recovery from illnesses, physical strain, and even psychological stress becomes hindered. At times, adults may never fully recover from challenges that would have been insignificant in their youth.
In childhood, the body operates with heightened plasticity due to the still-developing organs, neural connections, immune pathways, and metabolic systems. Since growth is the top priority, biological systems adapt to facilitate efficient repair mechanisms and high cellular turnover rate. As growth transitions into maintenance during adulthood, repair processes become less efficient, damage accumulates, and the margin for recovery narrows. Researchers in aging biology, such Dr. Svetlana Ukraintseva, a professor in the Social Sciences Institute at Duke University, describes aging to be a gradual erosion of recovery capacity that is driven by reserve depletion, imperfect repair, and a slowdown in physiological responses.
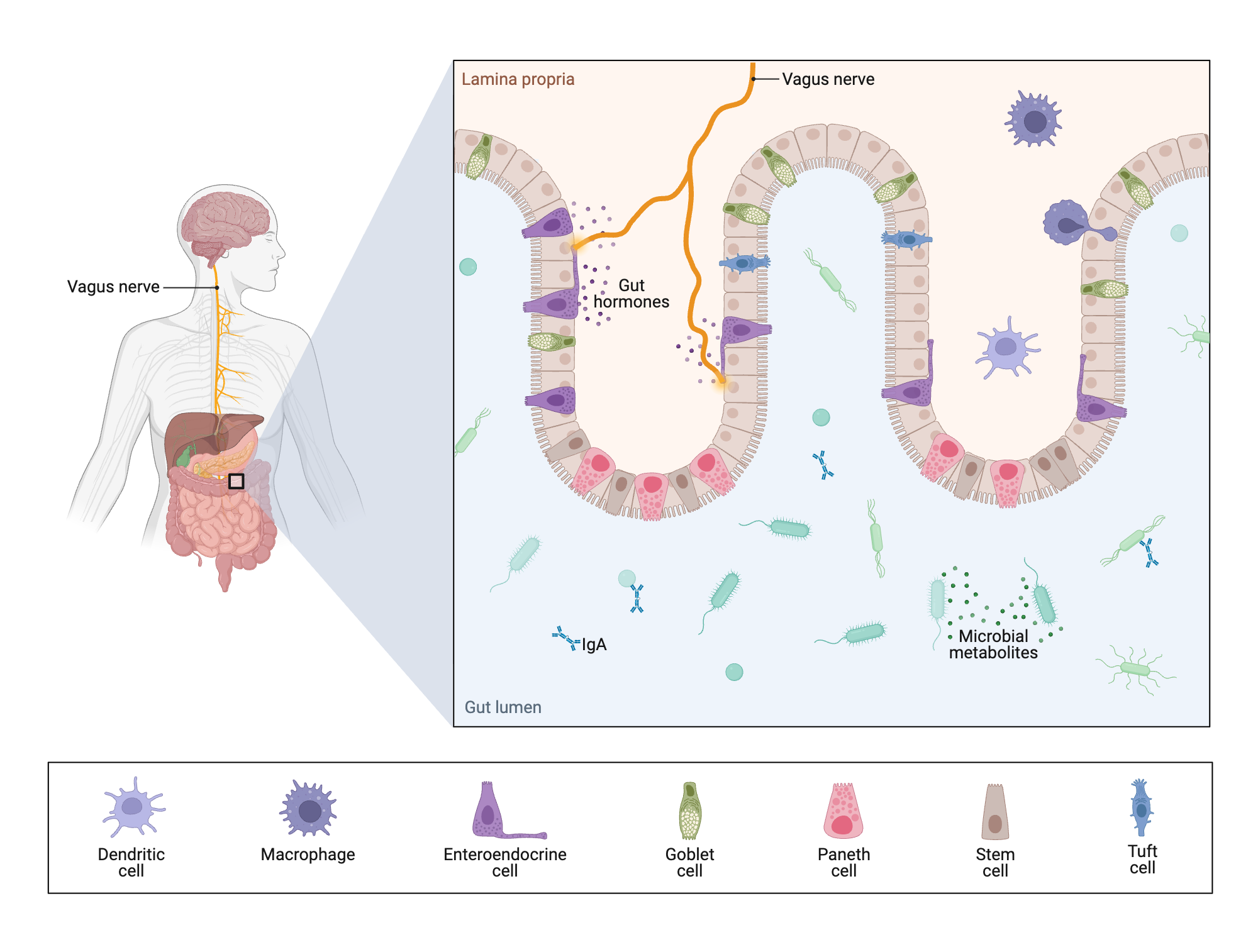
Graphic by Samara Ona using BioRender. Curated by Hailey Foster (hf348@cornell.edu).
Nonetheless, biological resilience can vary across children, as childhood is a period of biological calibration where early experiences shape stress-response systems. Drawing from the evolutionary principles articulated by Charles Darwin, developing organisms adjust their biology in response to environmental signals. Supportive environments calibrate stress systems towards a more balanced activation, while unpredictable environments might condition a child toward heightened vigilance. These adaptations seem to be mediated through epigenetic mechanisms such as DNA methylation, a type of chemical modification that regulates gene expression throughout the genome without changing DNA sequences. While such calibration can be adaptive in context, prolonged exposure to stress and heightened vigilance may lead to long-term physiological consequences.
Advances in molecular biology now allow researchers to track biological aging using epigenetic clocks based on DNA methylation patterns. Epigenetic clocks estimate biological age by studying predictable patterns of DNA methylation. Early clocks developed by researchers Steve Horvath and Gregory Hannum predicted chronological age from these patterns. Today, the newer tools of GrimAge and DunedinPACE are designed to directly estimate mortality risk and the pace of physiological aging. Evidence suggests that higher psychosocial resilience in childhood, exhibited through greater senses of focus, responsibility, and ability to cooperate, are associated with slower epigenetic aging in adulthood.
Photos from 100% PURE (left) and International Food Information Council (right). Curated by Hailey Foster (hf348@cornell.edu).
Biological resilience declines with age as the sum of metabolic strain, inflammation and repeated stresses erode the body’s recovery systems and slow physiological responses. Additionally, with age, DNA repair becomes less precise and mutations become increasingly likely. Evolutionary pressures favor survival and reproduction, but fail to favor maintenance of peak repair capacity later in life. Understanding biological aging reframes aging itself as not just growing older, but as the gradual loss of the body’s ability to restore balance. Recognizing such loss is necessarily the very first step in identifying novel ways to preserve and enhance biological resilience.
Photo from BCG Washington. Curated by Hailey Foster (hf348@cornell.edu).
Rathusha Nimalan ’28 is in the College of Arts and Sciences. She can be reached at rn349@cornell.edu.